Abstract
OBJECTIVE: In order to elucidate the possible mechanism on the proliferation of paroxysmal nocturnal hemoglobinuria (PNH) clone, and investigate the significance of gene mutation in PNH patients with thrombosis and provide a theoretical basis for related molecules markers and potential therapeutic targets in PNH, we performed the whole genome exon sequencing in PNH patients.
METHODS: The peripheral blood and nail specimens of 7 PNH and 6 AA-PNH patients were obtained and we have used V2's NimbleGen capture probe to capturethe whole exon group (WES), and performed whole genome sequencing of exon in patients, meanwhile, processing and analysis of sequencing data were obtained and compared with the 1000 genome project database, to obtain high frequency variation genes and further gene enrichment pathway analysis in these patients. Using a combination of KEGG pathway analysis and GO pathway enrichment analysis methods to analyze the experimental data.
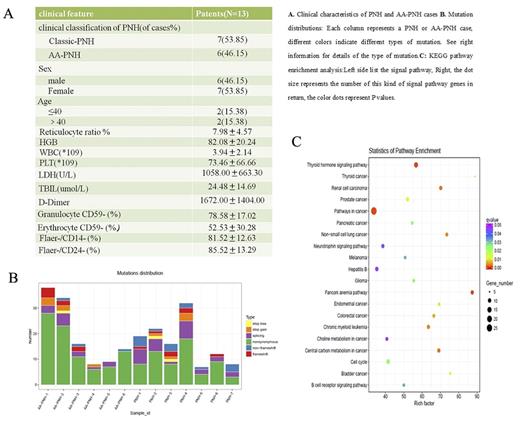
RESULTS: We detected somatic PIGA mutations in the PNH fractions of 8 of 13 cases analyzed by WES. And we identified a total of 4 shear site, 4 non-synonymous, 4 truncation, and 6 frame-shift mutations in cases analyzed by WES. All mutations were grade one. We performed KEGG pathway enrichment analysis and GO gene annotation of 5717 mutated genes screened, the union as a target gene set, there were 25 target genes, SUZ12, CUX1, MLL2, MUC4, CTBP1, AXIN2, MLL, SDHA, CARD11, MLH1, RB1, SMO and NKX2-1 mutations were recurrent in PNH patients; FGFR3, ERCC5, MPL, FLT3, TSC2, POLE, TET2, MLL3, MSH6, FAT1, CIC and ZFHX3 mutations were recurrent in AA-PNH patients. Among them, SUZ12, FLT3, CUX1, MUC4, TET2, and RB1 gene are involved in the regulation of cell proliferation and differentiation, promote the growth, metastasis and invasion of tumor cells. In addition, the pathways enriched by mutant genes included pathways in cancer, central carbon metabolism in cancer signaling pathway and cell cycle signal pathways. GO analysis showed that the coding protein CDP has the functions of cell proliferation regulation, metabolism regulation, gene expression regulation, cell invasiveness, cell motility, cell apoptosis, extracellular matrix compositions, and so on. Furthermore, we analyzed the gene mutations in patients with thrombosis, which includes several genes with the higher deleterious mutation in protein structure, such as BMPR2, ZFPM2, F5, F8, F12, F13A1, MCL1, MMRN1, NBEAL2, NOS1and PC. The major functions of mutation sites were non synonymous single nucleotide variants and synonymous single nucleotide variants. The pathways enriched by mutant genes included the Notch signaling pathway, Ribosome biogenesis in eukaryotes signaling pathway, Wnt signaling pathway and Arachidonic acid metabolism signaling pathways, which may be related to the pathogenesis of PNH associated thrombosis.
CONCLUSION: We identified additional somatic mutations that resulted in a complex hierarchical clonal architecture, similar to that observed in myeloid neoplasms and solid tumors. In addition to PIG-A mutations, mutations were found in genes known to be involved in myeloid neoplasm pathogenesis and solid tumors, including SUZ12, FLT3, CUX1, MUC4, TET2, and RB1. Our data indicate that in addition to PIG-A mutations, accessory genetic events are frequent in PNH, suggesting a stepwise clonal evolution derived from a singular stem cell clone. Meanwhile, several genes with higher deleterious mutation in PNH patients with thrombosis were found: F5, F8, F12, F13A1, THBD, ITGA2 and BMPR2, which enriched the Notch signaling pathway, Ribosome biogenesis in eukaryotes signaling pathway, Wnt signaling pathway and Arachidonic acid metabolism signaling pathways.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.